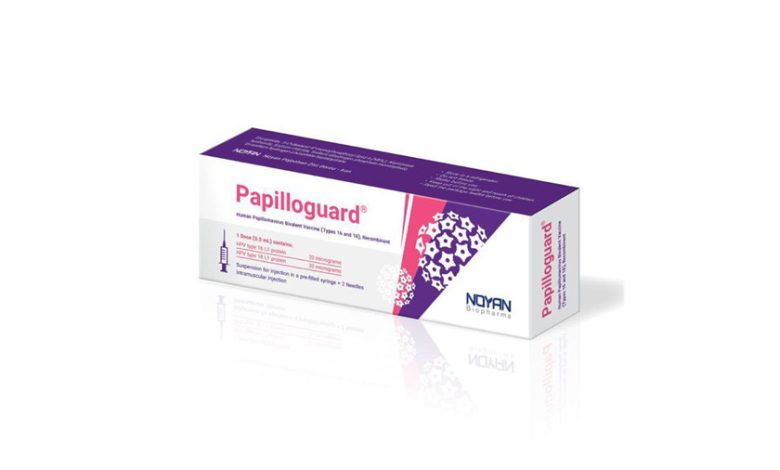
Papilloguard-4, the first Iranian HPV vaccine that had rapidly gained market dominance, was suddenly suspended by order of the Food and Drug Organization. Officials describe the decision as “purely administrative,” but for the public it raises concern and uncertainty. If the vaccine posed no problem, why was its production halted? While the Organization insists the issue was merely administrative, the pharmaceutical industry warns that such actions undermine public trust in domestic medicines.
Iranian Vaccine Halted at Peak Sales / Food and Drug Organization: “It Was an Administrative Issue, Not a Safety Concern!”
According to Salamat News, the sudden decision of the Legal Commission of the Food and Drug Organization to halt sales of the Iranian HPV vaccine “Papilloguard-4” has surprised not only pharmaceutical industry stakeholders but also the wider public. Developed by Nivad Pharmed Salamat, the vaccine was granted market authorization at the end of last year after extensive research and clinical trials, and within weeks secured a significant share of the domestic HPV vaccine market. Nevertheless, without any transparent explanation, a suspension order was issued. The company’s CEO has warned that this decision risks disrupting production cycles, damaging investor confidence, and undermining the future of Iran’s pharmaceutical industry.
In contrast, Dr. Mehdi Pirsolahi, head of the Food and Drug Organization, told Salamat News that the vaccine has no safety or quality issues, and that the suspension was solely for a renewed review of documentation. This contradiction leaves the central question unanswered: why was such an ambiguous decision made?
Background
In recent years, vaccination against Human Papillomavirus (HPV) has been recognized as one of the most effective tools in preventing cervical cancer and related diseases. Iran has sought to reduce dependency on imports by developing domestic production. Nivad Pharmed Salamat, under the brand name Papilloguard-4, succeeded in producing the country’s first quadrivalent HPV vaccine.
Following what experts describe as the largest clinical trial of its kind in Iran—conducted on more than 670 volunteers—the vaccine received approval on March 17, 2025. By April 4, it had reached pharmacies, and within weeks over 70,000 doses were sold, securing nearly 60% of the market—clear evidence of strong uptake among physicians and consumers.
But just as this growth accelerated, the Legal Commission of the Food and Drug Organization ordered a halt to sales—without any official statement, without clear explanation, and without details provided to the media or medical community.
Producer’s Objection: “Sudden Suspension Equals Distrust in Investment”
The CEO of Nivad Pharmed Salamat previously responded:
“No adverse events have been reported among recipients. All scientific and laboratory steps were conducted in line with the Food and Drug Organization’s standards. A sudden halt without clear explanation not only disrupts production but also undermines confidence in pharmaceutical investment. How can the future of this industry be assured if a product with official approval is suddenly removed from the market?”
This reflects a deeper concern: can investors in Iran’s domestic pharmaceutical sector rely on regulatory stability?
Regulator’s Response: “The Product Has No Issues”
Dr. Mehdi Pirsolahi, head of the Food and Drug Organization, offered a different account:
“Regarding the HPV vaccine, the suspension was neither due to product-related issues nor to any serious doubts about the documentation. The product has no problem, and still does not. The matter was simply that some documents and laboratory results needed re-examination in the Clinical Committee. The review is nearly complete, and the Commission’s renewed decision will be announced within days.”
He added that distributed vaccines were not recalled, meaning consumers can continue to use them. In other words, the Organization itself acknowledges no safety or quality concerns. The central question remains: if there were no problems, why was the suspension imposed?
Consequences of an Ambiguous Decision
While officials have attempted to frame the move as merely an “administrative review,” in public perception, the suspension of a vaccine immediately suggests doubts about its safety or effectiveness. Even if this interpretation is incorrect, the Organization’s silence and vague explanations fuel mistrust.
For the domestic pharmaceutical industry to grow and attract investment, regulatory predictability is essential. An investor who commits millions of dollars to research and vaccine production expects that, once approved, the product can enter the market without sudden, opaque risks. The suspension has not only caused financial and reputational harm to the manufacturer but also sent a discouraging signal to other industry players.
Beyond a Simple Administrative Action
The suspension of Papilloguard-4 is more than a routine administrative decision. It highlights one of the missing links in Iran’s healthcare policy: transparency in pharmaceutical regulation. While the head of the Food and Drug Organization insists the product is safe, the absence of timely and clear communication has proven nearly as damaging as an actual safety issue.
If Iran is to strengthen domestic production and pursue pharmaceutical self-sufficiency, the Food and Drug Organization must prioritize not only oversight but also public trust and accountability. More than ever, the domestic pharmaceutical sector needs transparency, regulatory stability, and precise communication—factors glaringly absent in the case of the HPV vaccine.